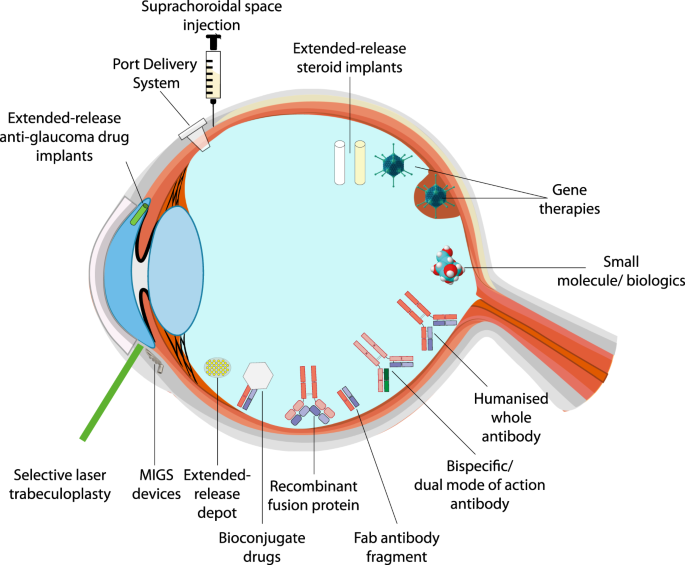
Pharmaceutics | Free Full-Text | Recent Advances of Ocular Drug Delivery Systems: Prominence of Ocular Implants for Chronic Eye Diseases

Effects of LATISSE (bimatoprost 0.03 per cent topical solution) on the ocular surface - Bitton - 2017 - Clinical and Experimental Optometry - Wiley Online Library
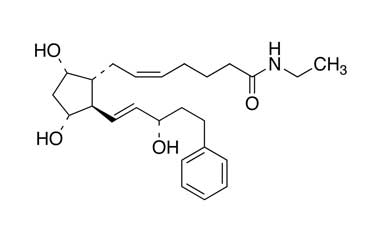
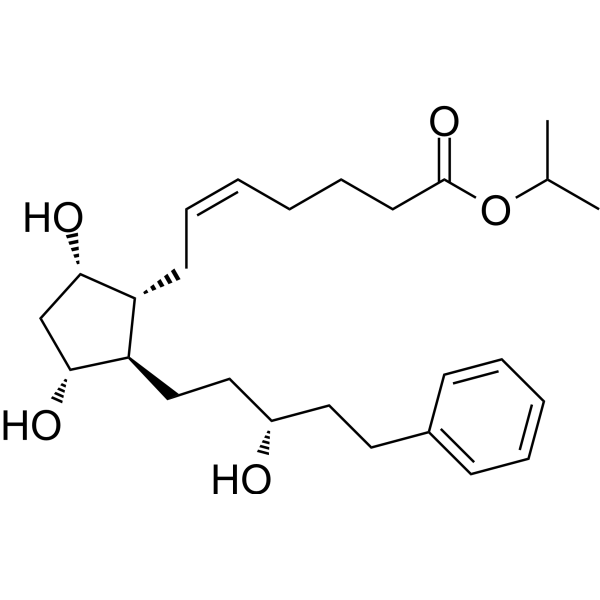
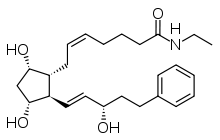
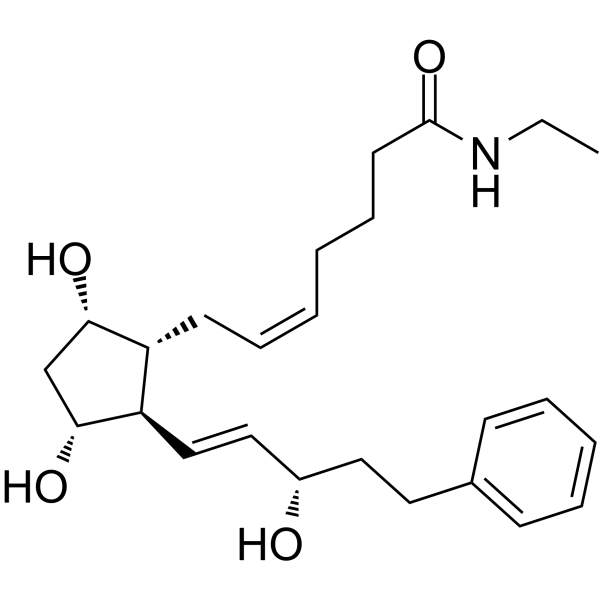
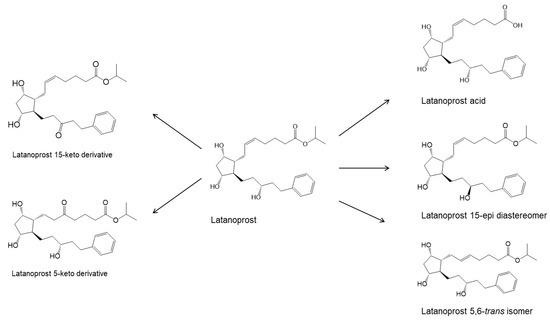
CAS No : 155206-00-1| Product Name : Bimatoprost - API| Chemical Name : Bimatoprost | Pharmaffiliates

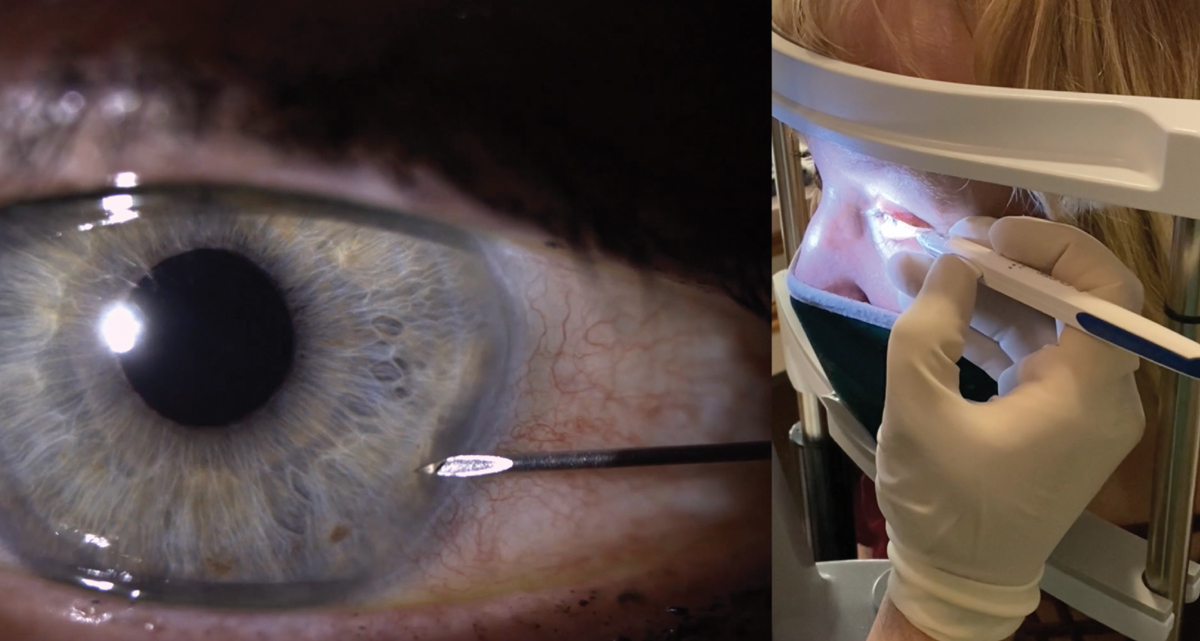
Allergan Receives FDA Approval for DURYSTA™ (bimatoprost implant) the First and Only Intracameral Biodegradable Sustained-Release Implant to lower Intraocular Pressure in Open-Angle Glaucoma or Ocular Hypertension Patients

Prolonged Ocular Retention of Mucoadhesive Nanoparticle Eye Drop Formulation Enables Treatment of Eye Diseases Using Significantly Reduced Dosage | Molecular Pharmaceutics

Effects of LATISSE (bimatoprost 0.03 per cent topical solution) on the ocular surface - Bitton - 2017 - Clinical and Experimental Optometry - Wiley Online Library
Public Assessment Report Scientific discussion Bimato-Vision 0.1 mg/ml, eye drops, solution (bimatoprost) NL/H/3025/001/DC Date

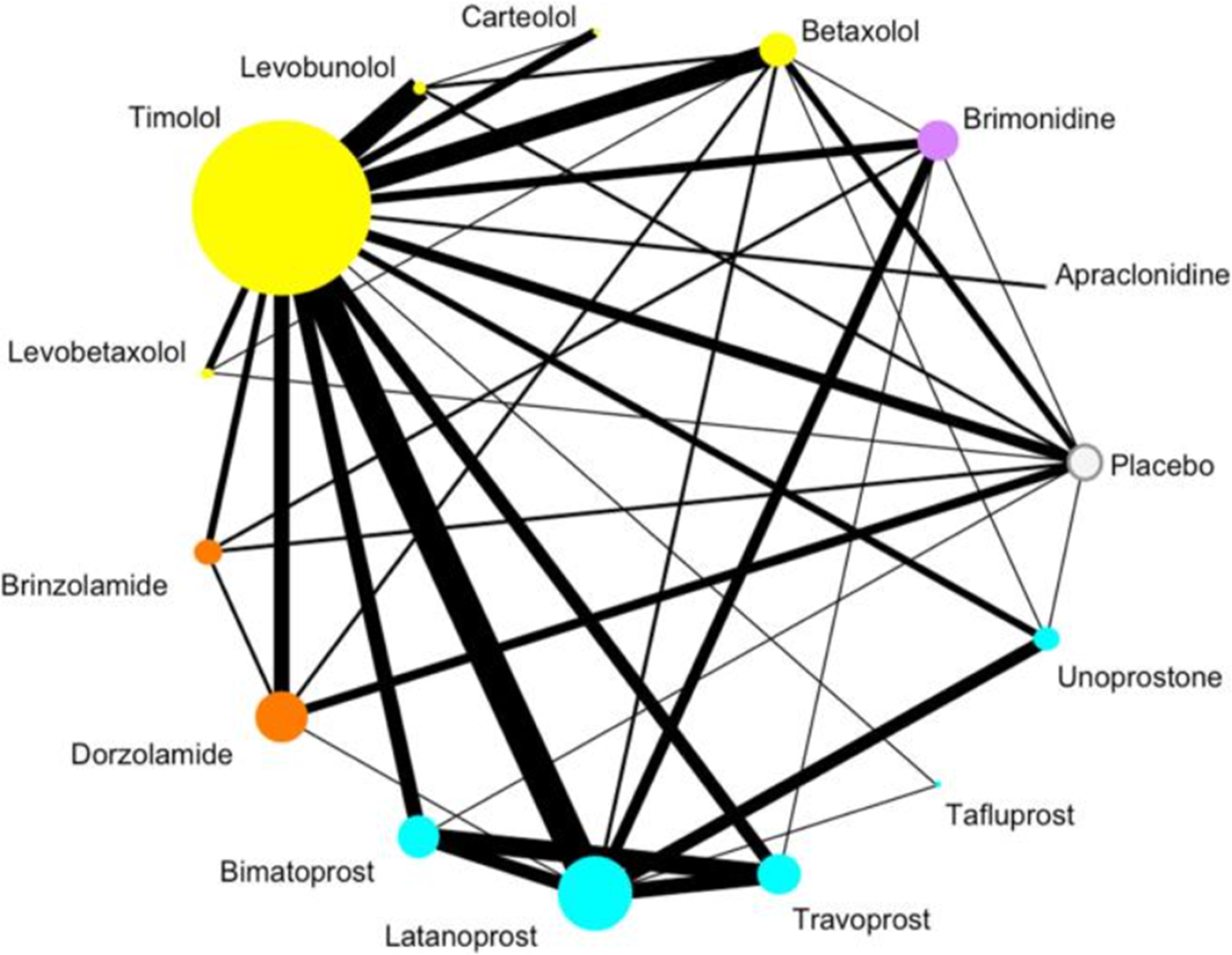
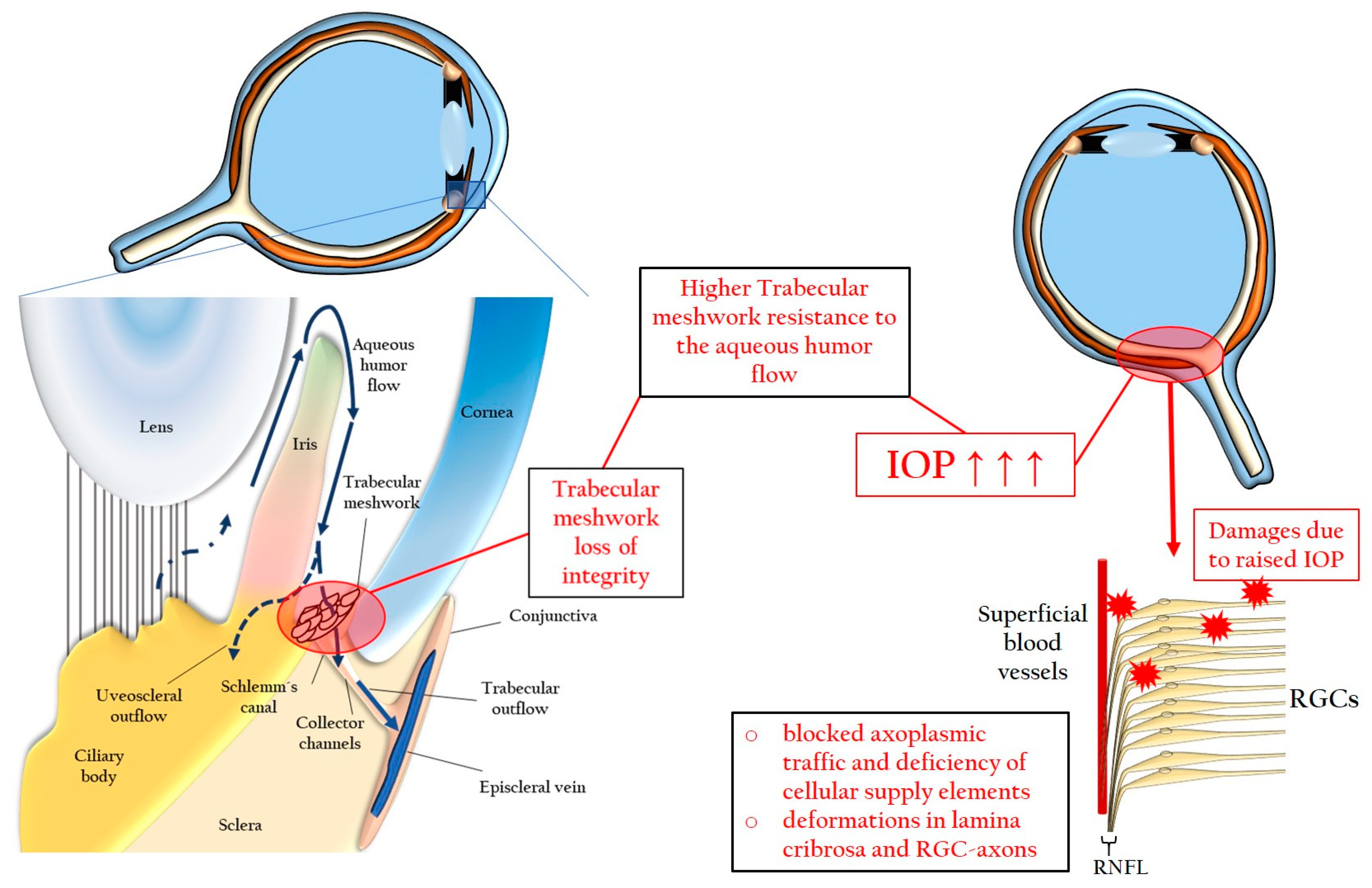
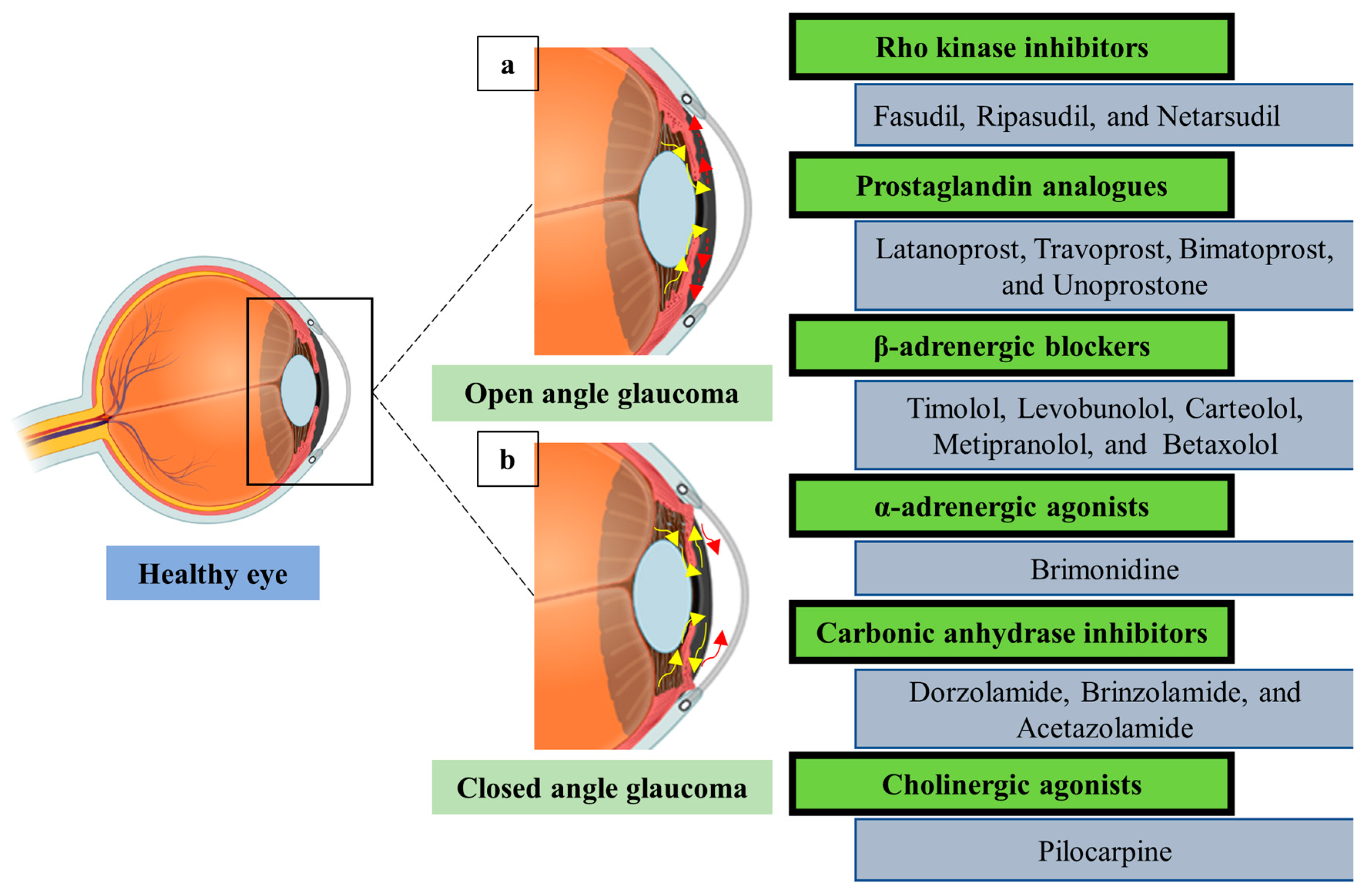
Medical anti‐glaucoma therapy: Beyond the drop - Miller - 2021 - Veterinary Ophthalmology - Wiley Online Library
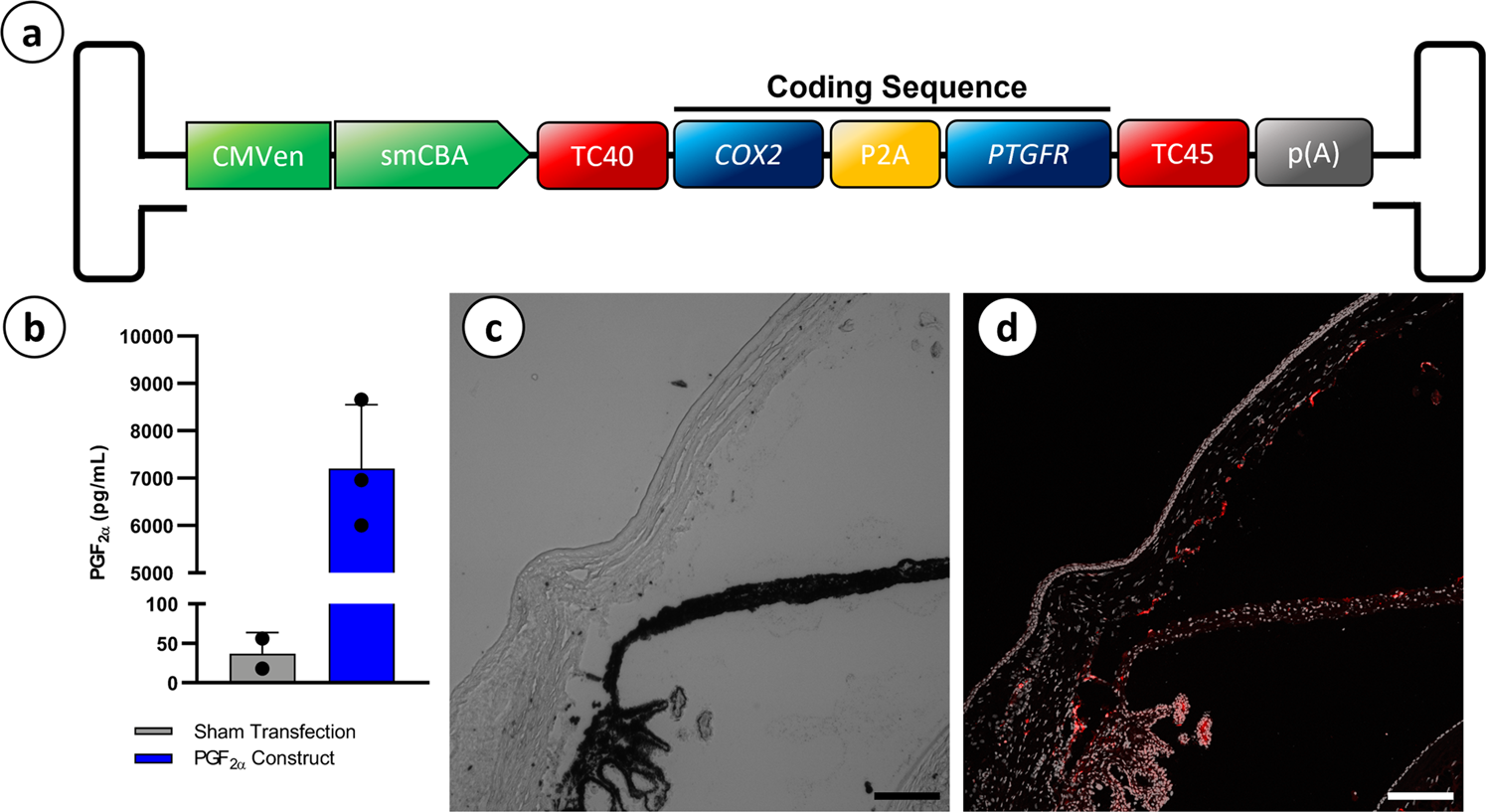
Prostaglandin-based rAAV-mediated glaucoma gene therapy in Brown Norway rats | Communications Biology

In situ gel containing Bimatoprost solid lipid nanoparticles for ocular delivery: In-vitro and ex-vivo evaluation - ScienceDirect

Impact of glaucoma medications on the ocular surface and how ocular surface disease can influence glaucoma treatment - ScienceDirect
Abstracts from the 2015 European Association for Vision and Eye Research Conference: Acta Ophthalmologica: Vol 93, No S255



![Latisse [5ml] – We Love Skin Latisse [5ml] – We Love Skin](http://weloveskinstore.com/cdn/shop/files/Latisse1.png?v=1691064420&width=1024)